Description
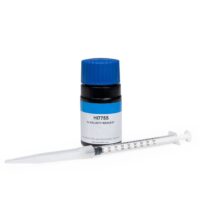
The HI84431-51 is a 100 mL bottle of acidic titrant solution that is used for high range (400 to 4000 mg/L as CaCO3) determination of alkalinity in water.
Water alkalinity is an expression of a water’s acid-neutralizing capacity and it is determined by titration with standard acidic solutions. Alkalinity is an important parameter for control and treatment of potable water and wastewater because it indicates the buffering capacity of water, or the ability to resist a pH change. The ability to resist large swings in pH results primarily from the presence of bicarbonates and carbonates. A low water alkalinity signifies that the water is susceptible to pH change, while a high alkalinity indicates that water is able to resist a major change of pH.
Alkalinity can be used to estimate levels of hardness or to determine the corrosive capacity of water. Alkalinity of surface water, quantified by mg/L as CaCO3 or meq/L, may be caused by carbonates, bicarbonates, hydroxides, phosphates, borates, silicates, or organic acid salts. Conventionally, the water alkalinity is approximated as sum of components. Three types of alkalinities exist together to form total alkalinity (TA):
TA = OH– + CO32– + HCO3–
OH– = hydroxide alkalinity
CO32– = carbonate alkalinity
HCO3– = bicarbonate alkalinity
TA = total alkalinity
Different tests can be performed to determine the quantity of different alkalinities present. According to the Standard Methods of Water and Wastewater Analysis, there are two options to determine water alkalinity: 1) Strong alkalinity determination by titration of the water sample with strong acid solution to 8.3 pH (known as phenolphthalein alkalinity), or 2) Total alkalinity determination by titration of the water sample with strong acid solution to 4.5 pH (known as bromcresol green alkalinity).
Replacement reagents required to operate the HI84431:
|
Low Range – 10 to 500 mg/L as CaCO3
|
High Range – 400 to 4000 mg/L as CaCO3
|
|
Pump calibration standard
|
HI84431-55M
|
HI84431-55M
|
|
Titrant solution
|
HI84431-50
|
HI84431-51
|